
Biochemistry and Molecular Biophysics
Lab Website
Publications (PubMed / NIH)
Research
Select Publications
Walker EC, Javati S, Todd EM, Matlam JP, Lin X, Bryant M, Krone E, Ramani R, Chandra P, Green TP, Anaya EP, Zhou JY, Alexander KA, Tong RS, Yuasi L, Boluarte S, Yang F, Greenberg L, Nerbonne JM, Greenberg MJ, Clemens RA, Philips JA, Wilson LD, Halabi CM, DeBosch BJ, Blyth CC, Druley TE, Kazura JW, Pomat WS, Morley SC. “Novel coenzyme Q6 genetic variant increases susceptibility to pneumococcal disease.” Nat Immunol. 2024 Dec;25(12):2247-2258. doi: 10.1038/s41590-024-01998-4. Epub 2024 Nov 4. PMID: 39496954 (Abstract)
Michael J Greenberg (2024). “Super Relaxed or Super Stressed: Modeling Length-Dependent Activation in Cardiac Muscle” Biophys J.2024 Jul 26:S0006-3495(24)00492-2. doi: 10.1016/j.bpj.2024.07.033. (Abstract)
Ankit Garg, Silvia Jansen, Rui Zhang, Kory J Lavine & Michael J Greenberg (2024). “Dilated cardiomyopathy-associated skeletal muscle actin (ACTA1) mutation R256H disrupts actin structure and function and causes cardiomyocyte hypocontractility” bioRxiv. [Preprint]. 2024 Mar 12:2024.03.10.583979. doi: 10.1101/2024.03.10.583979. (Abstract)
Jasmine Cubuk, Lina Greenberg, Akiva E Greenberg, Ryan J Emenecker, Melissa D Stuchell-Brereton, Alex S Holehouse, Andrea Soranno & Michael J Greenberg (2024). “Structural dynamics of the intrinsically disordered linker region of cardiac troponin T” bioRxiv. [Preprint]. 2024 May 31:2024.05.30.596451. doi: 10.1101/2024.05.30.596451. (Abstract)
Ankit Garg, Silvia Jansen, Rui Zhang, Kory J Lavine, & Michael J Greenberg (2024). “Dilated cardiomyopathy-associated skeletal muscle actin (ACTA1) mutation R256H disrupts actin structure and function and causes cardiomyocyte hypocontractility” bioRxiv [Preprint]. 2024 Mar 12:2024.03.10.583979. doi: 10.1101/2024.03.10.583979 (Abstract)
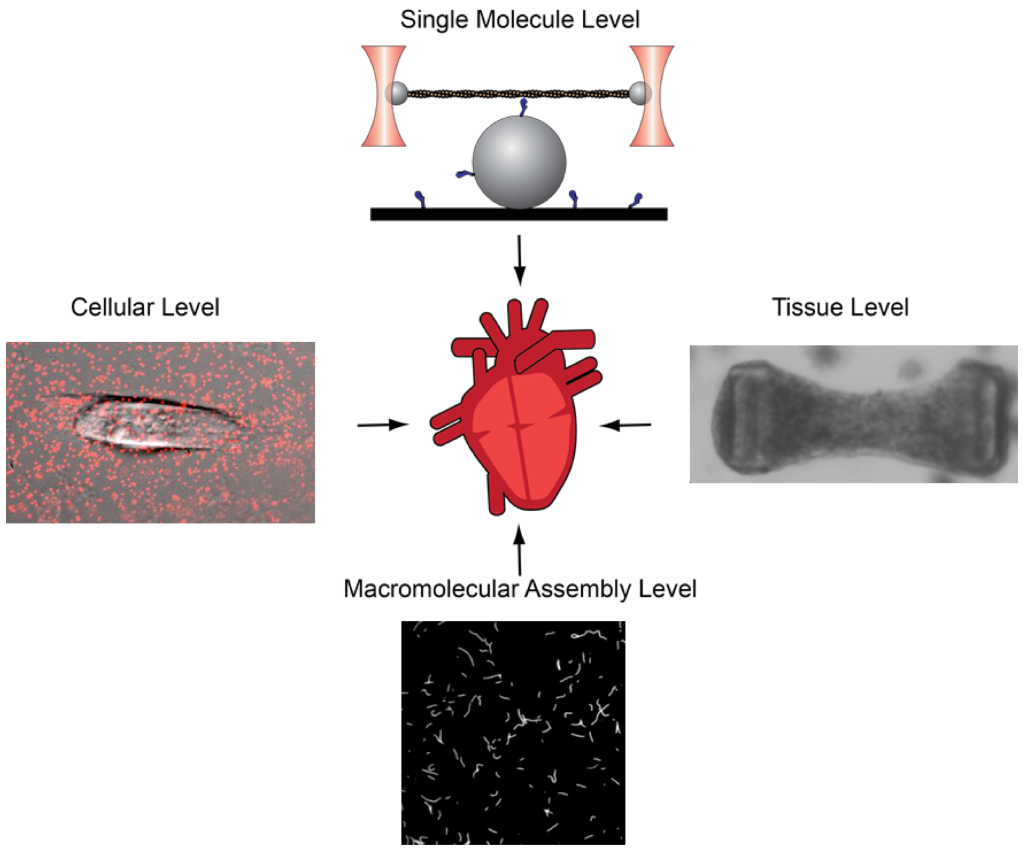
Ankit Garg, Kory J Lavine, & Michael J Greenberg (2024). “Assessing Cardiac Contractility From Single Molecules to Whole Hearts” JACC Basic Transl Sci. 2023 Oct 11;9(3):414-439. doi: 10.1016/j.jacbts.2023.07.013. eCollection 2024 Mar. (Abstract)
Gretchen A Meyer, Jeremie L A Ferey, James A Sanford, Liam S Fitzgerald, Akiva E Greenberg, Kristoffer Svensson, Michael J Greenberg, & Simon Schenk (2024). “Insights into post-translational regulation of skeletal muscle contractile function by the acetyltransferases, p300 and CBP” bioRxiv [Preprint]. 2024 Feb 29:2024.02.27.582179. doi: 10.1101/2024.02.27.582179. (Abstract)
Shahnaz Ghahremani, Aditya Kanwal, Anthony Pettinato, Feria Ladha, Nicholas Legere, Ketan Thakar, Yanfen Zhu, Harianto Tjong, Andrea Wilderman, W Tom Stump, Lina Greenberg, Michael J Greenberg, Justin Cotney, Chia-Lin Wei, & J Travis Hinson (2024). “CRISPR Activation Reverses Haploinsufficiency and Functional Deficits Caused by TTN Truncation Variants” Circulation. 2024 Apr 16;149(16):1285-1297. doi: 10.1161/CIRCULATIONAHA.123.063972. Epub 2024 Jan 18. (Abstract)
Brent Scott & Michael J Greenberg (2023). “Multiscale biophysical models of cardiomyopathies reveal complexities challenging existing dogmas” Biophys J. 2023 Dec 19;122(24):4632-4634. doi: 10.1016/j.bpj.2023.11.014. Epub 2023 Nov 25. (Abstract)
Samantha K. Barrick & Michael J. Greenberg (2021). “Cardiac myosin contraction and mechanotransduction in health and disease” J Biol Chem. 2021 Nov;297(5):101297. doi: 10.1016/j.jbc.2021.101297. Epub 2021 Oct 9. (Abstract)
Nicole R. Wong, Jay Mohan, Benjamin J. Kopecky, Shuchi Guo, Lixia Du, Jamison Leid, Guoshuai Feng, Inessa Lokshina, Oleksandr Dmytrenko, Hannah Luehmann, Geetika Bajpai, Laura Ewald, Lauren Bell, Nikhil Patel, Andrea Bredemeyer, Carla J. Weinheimer, Jessica M. Nigro, Attila Kovacs, Sachio Morimoto, Peter O. Bayguinov, Max R. Fisher, W. Tom Stump, Michael Greenberg, James A. J. Fitzpatrick, Slava Epelman, Daniel Kreisel, Rajan Sah, Yongjian Liu, Hongzhen Hu, & Kory J. Lavine (2021). “Resident cardiac macrophages mediate adaptive myocardial remodeling” Immunity. 2021 Sep 14;54(9):2072-2088.e7. doi: 10.1016/j.immuni.2021.07.003. Epub 2021 Jul 27. (Abstract)
Maria Papadaki, Theerachat Kampaengsri, Samantha K. Barrick, Stuart G. Campbell, Dirk von Lewinski, Peter P. Rainer, Samantha P. Harris, Michael J. Greenberg, & Jonathan A. Kirk (2021). “Myofilament glycation in diabetes reduces contractility by inhibiting tropomyosin movement, is rescued by cMyBPC domains” J Mol Cell Cardiol. 2021 Sep 3;162:1-9. doi: 10.1016/j.yjmcc.2021.08.012. Online ahead of print. (Abstract)
Samantha K. Barrick, Lina Greenberg, & Michael J. Greenberg (2021). “A Troponin T Variant Linked with Pediatric Dilated Cardiomyopathy Reduces the Coupling of Thin Filament Activation to Myosin and Calcium Binding” Mol Biol Cell. 2021 Jun 23;mbcE21020082. doi: 10.1091/mbc.E21-02-0082. Online ahead of print. (Abstract)
Sarah R. Clippinger, Paige E. Cloonan, Wei Wang, Lina Greenberg, W. Tom Stump, Paweorn Angsutararux, Jeanne M. Nerbonne, & Michael J. Greenberg (2021). “Mechanical dysfunction of the sarcomere induced by a pathogenic mutation in troponin T drives cellular adaptation” J Gen Physiol. 2021 May 3;153(5):e202012787. doi: 10.1085/jgp.202012787. (Abstract)
Michael J. Greenberg & Jil C. Tardiff (2021). “Complexity in genetic cardiomyopathies and new approaches for mechanism-based precision medicine” J Gen Physiol. 2021 Mar 1;153(3):e202012662. doi: 10.1085/jgp.202012662. (Abstract)
